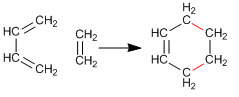
The Diels-Alder is a concerted reaction between a diene in the s-cis conformation and a dienophile -alkene-. The Diels-Alder products are cyclohexene derivatives.
Stereospecific Reaction
It is a stereospecific reaction; it preserves the stereochemistry of the diene and the dienophile and follows the endo rule.
Rich dienes and poor dienophiles
Rich dienes (with electron-donating substituents) react quickly with poor dienophiles (with electron-withdrawing substituents).
Electrocyclic reactions
Butadiene and 1,3,5-hexatriene undergo electrocyclic reactions that lead to concerted ring closures and openings.
Electrocyclic reactions can take place with light or heat and can be conrotatory or disrotatory.
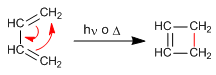
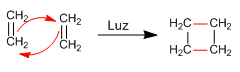
Photochemical reactions
In the presence of light, photochemical reactions occur. Alkenes join at the sp2 carbons, giving rise to four-membered rings.