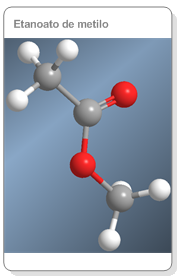
 Nomenclature of Esters
Nomenclature of Esters
They are named as alkyl alkanoates (methyl methanoate).
Properties of Esters
Esters give flavor and odor to many fruits and are the major constituents of animal and vegetable waxes.
Hydrolysis of Esters
The hydrolysis of esters is catalyzed by acids or bases and leads to carboxylic acids.
Transesterification
Esters react with alcohols under acid or base catalysis to yield a new ester without needing to go through the free carboxylic acid. This reaction is called transesterification.
Reaction with Organometallics
Grignard reagents transform esters into alcohols. The reaction cannot be stopped, and the addition of two equivalents of the organometallic occurs.
Reduction to Alcohols and Aldehydes
Lithium aluminum hydride transforms them into alcohols, and DIBAL into aldehydes.
Claisen Condensation
In basic media, they form enolates that condense, generating 3-ketoesters. This reaction is called the Claisen condensation.
Keep learning more about esters............