Rule 1. The IUPAC names acyl halides by replacing the -oic ending of the acid with the same number of carbons with -oyl. Additionally, the word acid is replaced with the corresponding halogen, named as a salt.
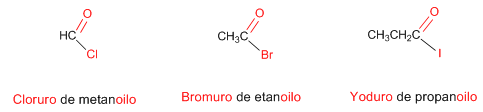
Rule 2. The longest chain containing the functional group is chosen as the principal chain. The numbering is done by assigning the lowest locator to the carbon of the halide.

Rule 3. This functional group takes priority over amines, alcohols, aldehydes, ketones, nitriles, and amides (which must be named as substituents). Only carboxylic acids, anhydrides, and esters have priority over it.

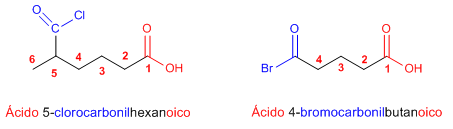
Rule 4. When there is a group in the molecule with higher priority than the halide (carboxylic acid, anhydride, ester), the halide is named as: halogenocarbonyl.......