Exercise 1.
The longest chain is selected as the main chain, such that it contains the largest number of substituents.
The longest chain is selected as the main chain, such that it contains the largest number of substituents.
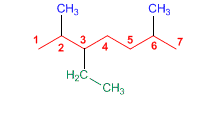
3-Ethyl-2,6-dimethylheptane
The substituents are an ethyl at position 3 and two methyls at positions 2 and 4. The substituents are arranged alphabetically, ethyl first then methyl. Quantity prefixes di, tri, etc., are not considered when alphabetizing.
Exercise 2.
The longest chain has 10 carbons, with two substituents at positions 4 and 8.
The longest chain has 10 carbons, with two substituents at positions 4 and 8.
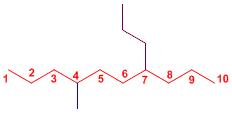
4-Methyl-7-propyldecane
The name of the molecule is constructed starting with the substituents, preceded by the locators that indicate the position they occupy within the main chain, and ends with the name of the main chain.
Exercise 3.
In this molecule, choosing the main chain can be misleading, as the horizontal chain only leads to 8 carbons, while the chain drawn in blue has 9 carbons.
In this molecule, choosing the main chain can be misleading, as the horizontal chain only leads to 8 carbons, while the chain drawn in blue has 9 carbons.
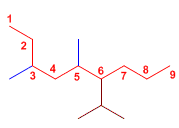
6-Isopropyl-3,5-dimethylnonane
The substituents are two methyl groups at positions 3,5 and an isopropyl group at position 6. The main chain of 9 carbons is named nonane.
Exercise 4.
The longest chain is taken as the main chain, 7 carbons.
The longest chain is taken as the main chain, 7 carbons.
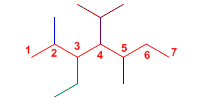
3-Ethyl-4-isopropyl-2,5-dimethylheptane
The molecule's substituents are an ethyl, an isopropyl, and two methyls. It is numbered from the left end so that the substituents take the lowest locators. The substituents are ordered alphabetically and the molecule's name ends in heptane.
Exercise 5
The main chain consists of nine carbons, therefore named nonane. The substituents are: ethyl, isopropyl, and methyl.
The main chain consists of nine carbons, therefore named nonane. The substituents are: ethyl, isopropyl, and methyl.
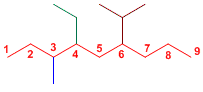
4-Ethyl-5-isopropyl-3-methylnonane
It is numbered from the left end, so that the locators are the lowest.
Exercise 6
The main chain is seven carbons: heptane.
The main chain is seven carbons: heptane.
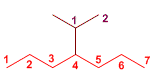
4-(1-Methylethyl)heptane
It has a complex substituent at position 4, named as methylethyl or (isopropyl).
Exercise 7.
The main chain has 10 carbons. It has substituents at C3, C4, C6, and C9.
The main chain has 10 carbons. It has substituents at C3, C4, C6, and C9.
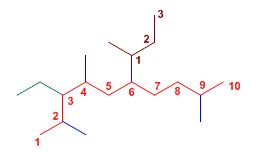
3-Ethyl-2,4,9-trimethyl-6-(1-methylpropyl)decane
At C6, it has a complex substituent, we number the secondary chain and name it as 1-methylpropyl.
Exercise 8.
The longest chain is taken as the main chain (in red).
The longest chain is taken as the main chain (in red).
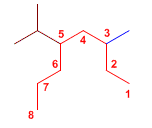
5-Isopropyl-3-methyl octane
It is numbered starting from the right, so we find the methyl at position 3 and the isopropyl at position 5.
Exercise 9.
Main chain of 6 carbons, with three methyls at 2,2,4 and an ethyl at 4.
Main chain of 6 carbons, with three methyls at 2,2,4 and an ethyl at 4.
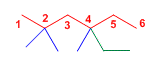
5-Ethyl-2,2,4-trimethylhexane