Cyclopropane Structure
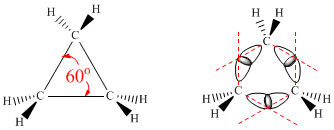
The C-C-C bond angle in cyclopropane is 60º, which is much smaller than the natural angle of a C(sp3) bond, which is 109.5º. In practice, the C-C bonds in cyclopropane bend outwards to allow overlap, resulting in longer and weaker bonds than those in normal alkanes. Another factor contributing to the strain in cyclopropane is hydrogen eclipsing.

Cyclopropane is a planar molecule with six eclipsed hydrogens and significant angular strain. The sigma carbon-carbon bonds are slightly bent outwards due to the significant strain. These curved bonds are called banana bonds.
Video
Additional Notes
* Cyclopropane is a highly reactive molecule due to its strained structure.
* Cyclopropane is readily halogenated, added across double bonds, and undergoes ring-opening reactions.
* Cyclopropane is found in small amounts in petroleum.