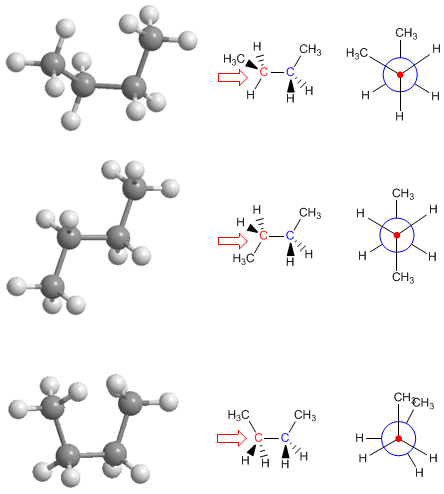
 What is a Newman Projection?
What is a Newman Projection?
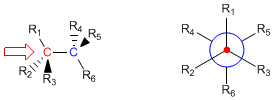
A Newman projection is obtained by looking at the molecule along the C-C axis. The front carbon is represented by a point, from which three bonds extend to its substituents. The rear carbon is represented by a circle, and the bonds emerging from this carbon are drawn starting from the circle.
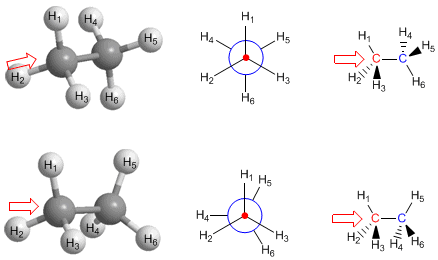
Newman Projection of Staggered and Eclipsed Ethane
The conformations of ethane can be easily represented using this projection. In the case of the eclipsed conformation, to make the three rear hydrogens more visible, they are drawn slightly rotated from the fully eclipsed position.
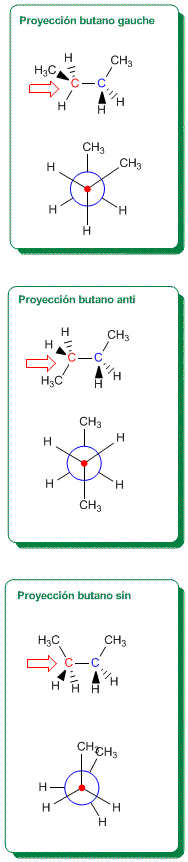
Newman Projection for the Conformations of Butane
The right column includes the syn, anti, and gauche conformations of butane, with their respective Newman projections, where the interactions between methyl groups can be observed. The most stable conformation is the anti (no methyl-methyl repulsions), with the syn conformation being the most unstable (high energy) due to methyl-methyl eclipsing.