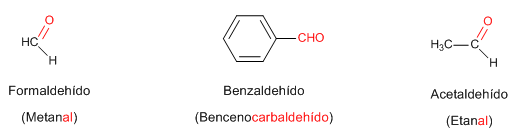
Rule 1. Aldehydes are named by replacing the -e ending of the corresponding alkane with -al. It's not necessary to specify the position of the aldehyde group since it occupies the end of the chain (locator 1).
When the chain contains two aldehyde functions, the suffix -dial is used.
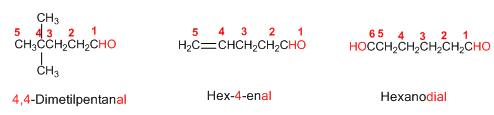
Rule 2. The -CHO group is named -carbaldehyde. This type of nomenclature is very useful when the aldehyde group is attached to a ring. The numbering of the ring is done by giving locator 1 to the ring's carbon that contains the aldehyde group.

Rule 3. When there is a group in the molecule with higher priority than the aldehyde, it becomes a substituent named as oxo- or formyl-.
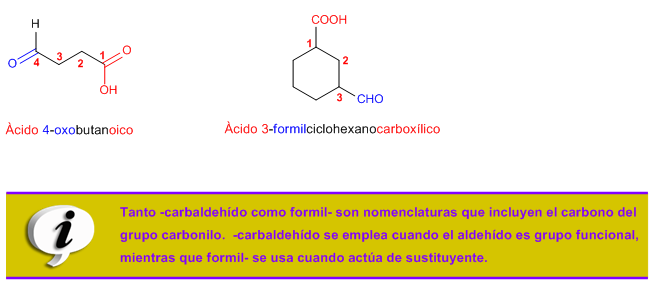
Rule 4. Some common names of aldehydes accepted by IUPAC are: