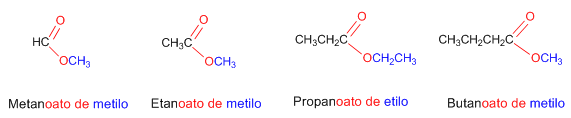
Rule 1. Esters result from the condensation of acids with alcohols and are named as salts of the acid they derive from. The IUPAC nomenclature changes the -oic ending of the acid to -oate, ending with the name of the alkyl group attached to the oxygen.
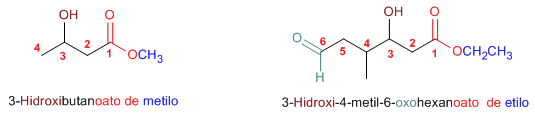
Rule 2. Esters are priority groups over amines, alcohols, ketones, aldehydes, nitriles, amides, and acyl halides. These groups are named as substituents with the ester being the functional group.
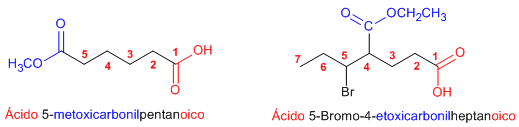
Rule 3. Carboxylic acids and anhydrides have priority over esters, which are then named as substituents (alkoxycarbonyl......).
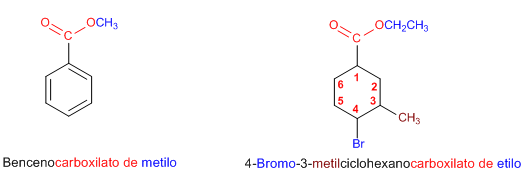
Rule 4. When the ester group is attached to a cycle, the cycle is named as the main chain and the ester is named using the -alkyl carboxylate ending.